Background:
Acute myeloid leukemia (AML) and myelodysplasia (MDS) are usually associated with poor outcomes, especially in high-risk AML/MDS. Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only curable option for patients suffering from high-risk AML/MDS. However, there were still many patients relapsed after allo-HSCT, especially for some patients are MRD positive before transplantation. Novel therapy to prevent replase is urgently needed. Both BCL-2 inhibitor, venetoclax (VEN) and hypomethylating agent, decitabine (DEC) possess significant antitumor activity effects against AML/MDS. Administration DEC has been shown to ameliorate GVHD and boost GVL post-transplantation. Several clinical trials have also shown that venetoclax plus decitabine can be a safety and effective salvage treatment for patients with AML/MDS relapsing after allo-HSCT. We therefore conducted a prospective study (ChiCTR1900025374) to exam the tolerability and efficacy of a maintenance therapy low-dose decitabine (LDEC) plus VEN to prevent relapse after allo-HSCT for MRD positive high-risk AML/MDS patients. To our knowledge, this is the first report of venetoclax combined decitabine in this setting.
Methods:
Six patients with MRD positive high-risk AML (n=5) /MDS(n=1) post transplantation were recruited. Around day 100 post transplantation, all patients received LDEC (15mg/m2 for 3 days) followed by VEN (200mg) on day 1 to 21. Two months is a cycle. The primary end points of this study were rates of Overall survival (OS) and event-free survival (EFS). The secondary endpoints included adverse events (AEs), incidence of cumulative incidence of relapse (CIR), nonrelapse mortality (NRM), incidences of acute GVHD (aGVHD) and chronic GVHD (cGVHD) and incidences of viral infection after allo-HSCT. Survival outcomes were analyzed using Kaplan-Meier analysis
Results:
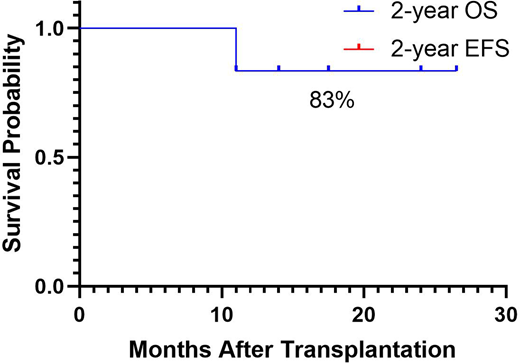
Two of the six patients were partial remission (PR) before transplantation, and the remaining 4 patients were MRD+ before transplantation. The median follow-up was 16 (11-26.5) months. Both 2-year OS and 2-year EFS were 83%. The median 2-year EFS time was 16(9-26.5) months, and five patients still EFS alive at the time of this writing. The 2-year cumulative incidence of relapse after LDEC+VEN was 17% and 2-year non-relapse mortality was 0%.
No tumor lysis syndrome (TLS) was observed. The most common AEs were neutropenia, anemia, thrombocytopenia, neutropenic fever, and fatigue. Grade 2 or 3 adverse events were observed in 33% (2/6). No grade>3 AEs were observed. Acute (any grade) and chronic (limited or extensive) graft-versus-host disease occurred in 67% and 17% of patients, respectively. The 2-year cumulative incidence of CMV viremia and EBV viremia were 33.3% and 16.7%, respectively.
Conclusion:
We conclude LDEC+VEN can be administered safely after allo-HSCT, without evidence for increased incidence of GVHD, and this combination demonstrates decreased relapse for MRD positive high-risk AML/MDS patients. This novel maintenance therapy may be a promising way to prevent replase for MRD positive high-risk AML/MDS patients, and the clinical benefits need to be assessed in a comparative prospective trial.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal